- No Results
- Global
-
Australia

-
Austria

-
Azerbaijan

-
Brazil

-
Belgium

-
Canada

-
Chile

-
China

-
Costa Rica

-
Croatia

-
Czech Republic

-
Denmark

-
ESTONIA

-
Finland

-
France

-
Germany

-
Hong Kong

-
Hungary

-
India

-
Italy

-
Ireland

-
Japan

-
Korea

-
Latvia

-
Lithuania

-
Malaysia

-
Mexico

-
Morocco

-
Netherlands

-
New Zealand

-
Norway

-
Philippines

-
Poland

-
Portugal

-
Romania

-
Singapore

-
Slovakia

-
Slovenia

-
Spain

-
Sweden

-
Switzerland

-
Taiwan

-
Turkey

-
United Kingdom

-
UNITED ARAB EMIRATES

-
United States

-
Vietnam

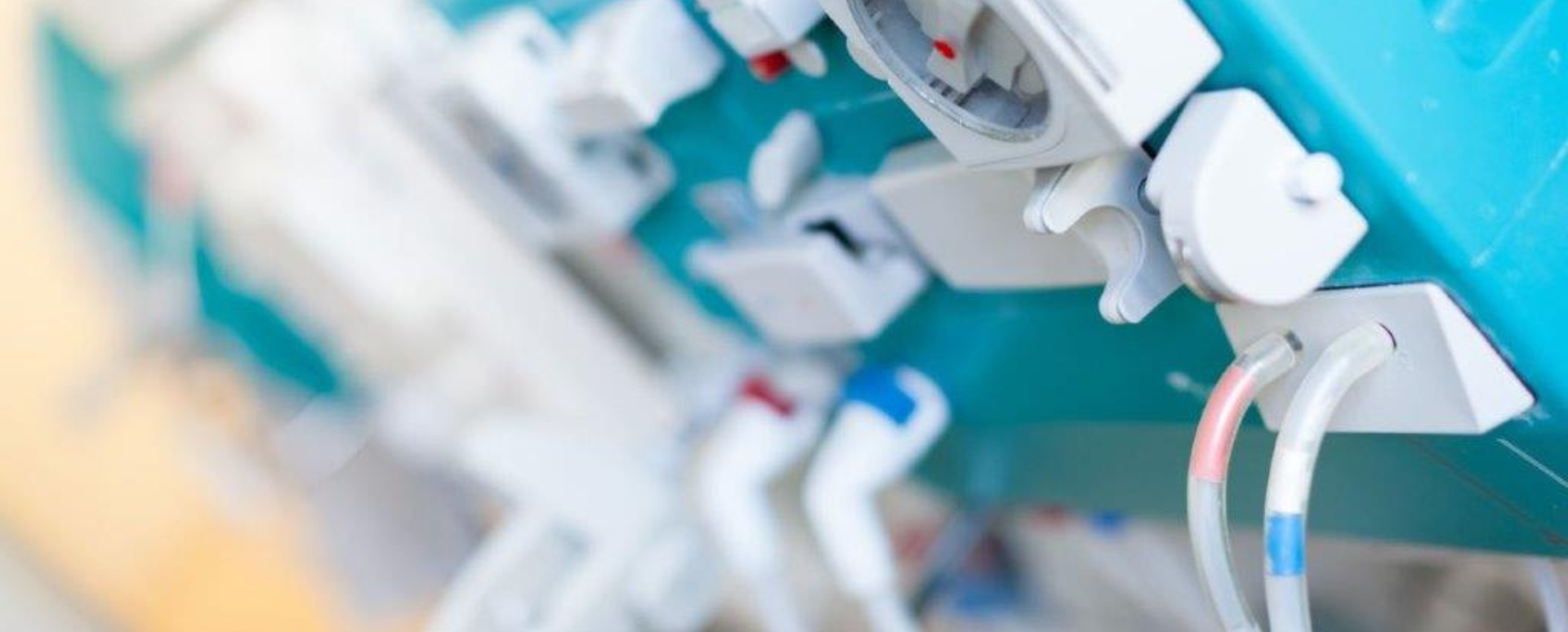
Electrical Medical Equipment
Contact usWorldwide medical electrical equipment and in vitro diagnostic medical devices are controlled by strictly regulated national and international approval procedures which include testing, certification and audit requirements.
Both current and upcoming regulations place a focus on evaluating and reducing the risks involved with medical devices and their connections to other devices and/or IT networks.
Eurofins Medical Device Testing laboratories and certification services can help you ensure compliance with the latest standards and regulations, while achieving Global Market Access (GMA) in a cost and time efficient manner.
Choose Eurofins Medical Device Testing to help you:
- Check basic safety and essential performances
- Achieve compliance with the Medical Device Regulation (MDR) 2017/745
- Achieve compliance with the Medical Device Directive (MDD) 93/42/EEC
- Achieve compliance with the In Vitro Diagnostic Medical Devices Directive (IVDD) 98/79/EC
- Ensure conformity to IEC 60601 & IEC 61010 standards*
- Obtain CB Scheme test reports and certificates
- Achieve ISO 13485
- Gain Global Market Access; including Europe (CE Mark) & US (Eurofins NRTL MET Mark) & Canada (SCC accreditation certification)
- Ensure your medical device is decure
*Where IEC standards are referred to, national and regional variations are also applicable including, but not limited to; ANSI/AAMI, UL, ISO, CAN/CSA, JIS, EN, ENV, BS EN, DD ENV and BS IEC. Please enquire on a case-by-case basis.
| Testing Services for Medical Electrical Equipment | ||
| Safety & Performance Testing | ||